Therefore, Cu could not possibly reduce H and this reaction would not take place. Some metals are more reactive than others. Related Byte: Redox Reaction Basics How To Determine Oxidation Number, Oxidizing and Reducing AgentsĪ: Copper because its charge increased from 0 to +2.Ī: H was reduced, making it an oxidizing agent (it oxidized Cu).Ī: Cu was oxidized, making it a reducing agent (it reduced H).Ī: No Since Cu is listed below H in the activity series, it means that hydrogen is actually the stronger reducer. Therefore, it makes sense that Mg would be the reducing agent. A single replacement reaction, sometimes called a single displacement reaction, is a reaction in which one element is substituted for another element in a compound. Zinc, lead, copper, and silver are placed in test tubes containing solutions of. Q: Does this reaction make sense according to the activity series?Ī: Yes Since Mg is listed above H in the activity series, it makes a stronger reducer. Activity Series of Metals Demonstration A 4X4 matrix of test tubes is set up. 
This is what determines if a reaction will or will not occur.Ī: Magnesium because its charge increased from 0 to +2.Ī: Hydrogen because its charge decreased from +1 to 0.Ī: H was reduced, making it an oxidizing agent (it oxidized Mg).Ī: Mg was oxidized, making it a reducing agent (it reduced H). A few drops of universal indicator have been added to the water.The activity series lists elements in their order of strength as oxidizers or reducers. Mnemonicate 04:56 Activity Series of Metals and Reaction Prediction. Questionĭescribe and explain the observations when a small piece of lithium is placed on the surface of a big container of water. In general, the more reactive the metal, the more rapid the reaction is. Along the side of the matrix, you should list the five metals used in this part of the experiment: copper, iron, magnesium, tin, and zinc. Sodium + water → sodium hydroxide + hydrogen For example, sodium reacts rapidly with cold water: Overview:The students will develop their own activity series of metals based on lab results. When a metal reacts with water, a metal hydroxide and hydrogen are formed. Hydrogen and carbon are shown for comparison. In a solution or compound, an elemental metal will. The reactivity series typically includes the following elements: potassium, sodium, calcium, magnesium, aluminum, zinc, iron, lead, copper, and silver. The table summarises some reactions of metals in the reactivity series. Organizing the metals in this fashion also allows us to predict how the metals will react with each other. the more easily it loses electrons in reactions to form positive ions (cations).+ Experiment number 2: To determine if the various metals will react with hydrochloric acid. 
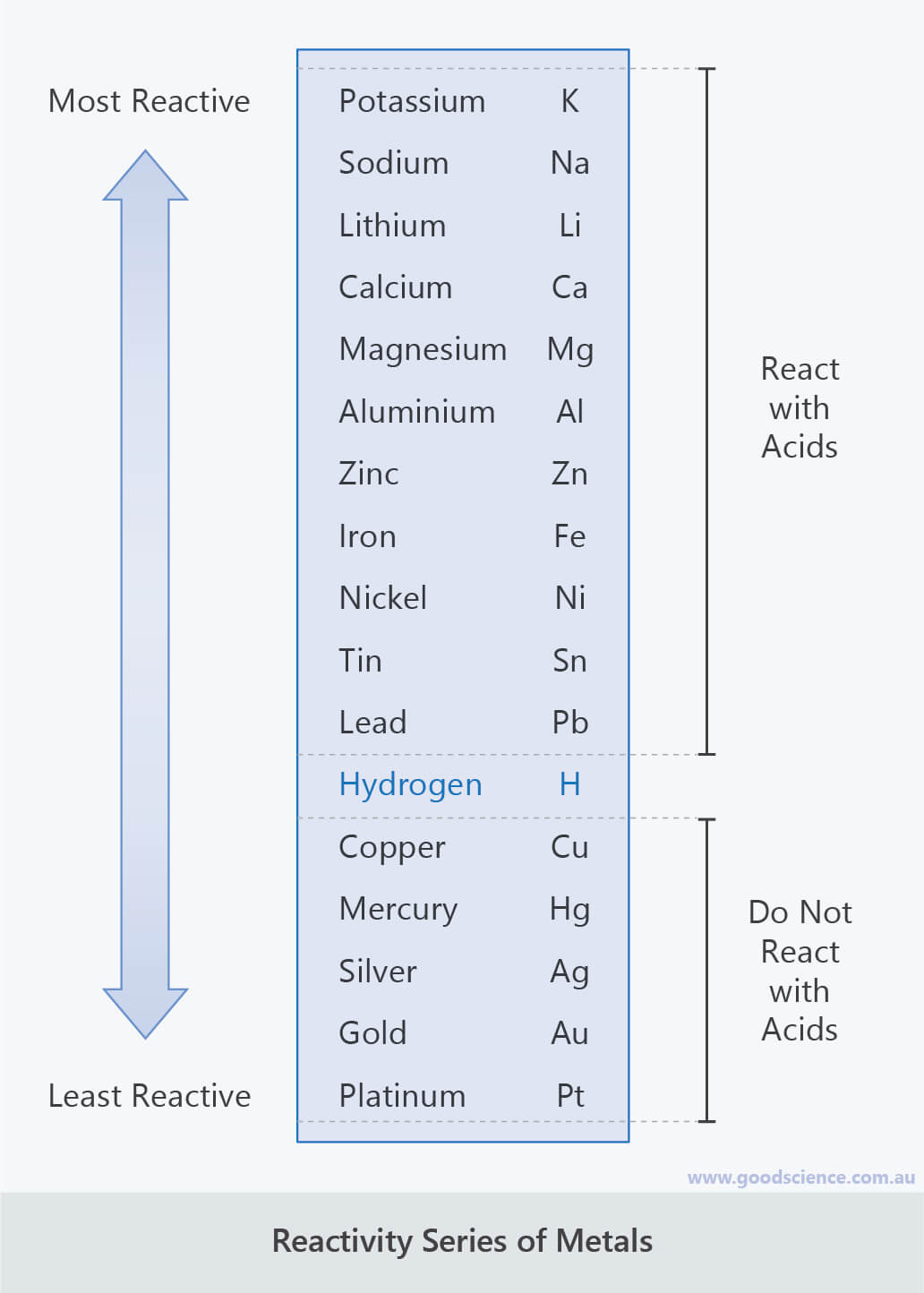
The easier it is for a metal to form its positive ion, the more reactive. In this lab, there will be 3 experiments: + Experiment number 1: To determine if the various metals will react with water. In general, the more reactive a metal is: When metals react with other substances, the metal atoms always form positive ions. 
The reactivity series of metals is a chart showing metals in order of decreasing reactivity. The activity series is a list of metals and their half-reactions arranged in order of decreasing ease of oxidation or increasing ability to take an electron. When metals react with other substances, the metal atoms lose electrons to form positive ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
